Dercum´s disease - a frequently
overlooked disease picture:
J. Steiner, K. Schiltz, F.
Heidenreich, K. Weissenborn (2002) NERVENARZT 73: 183-187
Neurologische Klinik, Medizinische Hochschule Hannover, Carl-Neuberg-Strasse 1, 30625 Hannover
Neurologische Klinik II, Otto-von-Guericke Universität Magdeburg, Leipziger Str. 44, 39120 Magdeburg
Abstract
Dercum's disease (lipomatosis dolorosa) is a relatively unknown illness. The disorder usually affects middle-aged females. Subcutaneous fatty tissue deposits may occur in many parts of the body. The upper arms, elbows, stomach wall, buttocks, thighs, or knees are most commonly affected. Severe hyperalgesia is found on light pressure and touch. Analgesics or pain-modulating drugs usually have little or no effect. The following case report demonstrates successful symptomatic treatment of the otherwise nearly unbearable complaints: intravenous infusions of 5 mg/kg body weight of lidocaine over 30–90 min may give pain relief lasting several weeks or even months. Alternatively, patients are treated with 150–750 mg orally administered mexiletine daily. Surgical excision or liposuction of these fatty tissue deposits have shown significant reduction of pain. However, this effect reduces over time and recurrences often develop.
___________________________________________________________________________
Dercum's disease (lipomatosis dolorosa) is a relatively unknown illness, although it was first described by the American neurologist Francis Xavier Dercum (1856 – 1931) in 1888 [9, 10]. A number of synonyms can be found in the medical specialty literature, including adiposalgia, adiposis dolorosa, adipositas dolorosa, Dercum's disease, adipose tissue rheumatism (Sweden), lipalgia or neurolipomatosis. WHO has assigned the ICD-10 number E88.20 to the syndrome.
Because of the painful subcutaneous fat deposits associated with the syndrome, this substantially dermatological pathology is of relevance to neurologists and pain therapists. The symptoms are virtually unaffected by conventional analgesics or classical pain modulation therapy (e.g. amitriptyline, carbamazepine). A better understanding of the characteristic syndrome would assist in early diagnosis and avoidance of unnecessary treatment attempts. The purpose of this article is, therefore, to introduce the current state of knowledge and opportunities for promising pain therapy.
Case report
A 47 year old female patient noticed for the first time in the late 80s multiple nodules on the inner surface of both upper arms; later 2 deep nodules on the left inner thigh were also noticed. When, in 1990 pain on contact and pressure occurred at the skin changes, the patient presented at a general practitioner, who informed her that it were lipomas – lipomas, however, do not cause pain.
 Nevertheless, the stress of the discomfort resulted in removal of
15 of these tumors in 1992. The histology of these tumors was
characteristic of lipoma and in part also angiolipoma. In the ensuing
years lipomas recurred, predominantly on the inner surface of the upper
arm and in the proximity of the elbow joint (Fig. 1), which were again
tender and sensitive to pressure. The symptoms were aggravated in winter
and at emotional stress. The patient intermittently complained of tingling
paresthesia at the fingertips accompanied by grip impairment. In addition,
there was a predisposition to ecchymoses. Since the patient complained of
occasional swelling of the fingers and dryness of the mouth lasting for
days, provisional pain therapy with meloxicam (Mobec®) was instituted on
the presumption of rheumatoid disease. The medication was later changed to
tramadol (Tramal®). However, neither of the two medications was effective.
In 1998 pain had reached such a level that it interfered with the
patient's sleep and she could no longer rest the arms on the torso. It was
at this time that the patient presented in our department for the first
time; initially on the presumption of a neurofibromatosis. The
neurological examination was unremarkable except for a painful limitation
of movement in both arms. Café-au-lait spots were not observed and the
cranial and cervical MRI studies were unremarkable – in
particular, neurofibromas were not demonstrated. At laboratory testing all
routine parameters were within normal, ESR 16 / 42 normal; RF,
ANA, cANCA and pANCA were negative. A Raynaud phenomenon with typical
cold-induced discoloration of the distal parts of the extremities
(tricolor phenomenon) was excluded in the differential diagnosis.
Electroneurography also did not provide evidence of the presence of a
sulcus-ulnaris or a carpal tunnel syndrome. Based on the characteristic
constellation of findings, we made the diagnosis of Dercum´s disease.
Nevertheless, the stress of the discomfort resulted in removal of
15 of these tumors in 1992. The histology of these tumors was
characteristic of lipoma and in part also angiolipoma. In the ensuing
years lipomas recurred, predominantly on the inner surface of the upper
arm and in the proximity of the elbow joint (Fig. 1), which were again
tender and sensitive to pressure. The symptoms were aggravated in winter
and at emotional stress. The patient intermittently complained of tingling
paresthesia at the fingertips accompanied by grip impairment. In addition,
there was a predisposition to ecchymoses. Since the patient complained of
occasional swelling of the fingers and dryness of the mouth lasting for
days, provisional pain therapy with meloxicam (Mobec®) was instituted on
the presumption of rheumatoid disease. The medication was later changed to
tramadol (Tramal®). However, neither of the two medications was effective.
In 1998 pain had reached such a level that it interfered with the
patient's sleep and she could no longer rest the arms on the torso. It was
at this time that the patient presented in our department for the first
time; initially on the presumption of a neurofibromatosis. The
neurological examination was unremarkable except for a painful limitation
of movement in both arms. Café-au-lait spots were not observed and the
cranial and cervical MRI studies were unremarkable – in
particular, neurofibromas were not demonstrated. At laboratory testing all
routine parameters were within normal, ESR 16 / 42 normal; RF,
ANA, cANCA and pANCA were negative. A Raynaud phenomenon with typical
cold-induced discoloration of the distal parts of the extremities
(tricolor phenomenon) was excluded in the differential diagnosis.
Electroneurography also did not provide evidence of the presence of a
sulcus-ulnaris or a carpal tunnel syndrome. Based on the characteristic
constellation of findings, we made the diagnosis of Dercum´s disease.
In November 1998, January 2000, July 2000, October 2000 and May 2001 systemic doses of lidocaine were administered. Initially, a test dose of 100 mg of lidocaine was administered i.v. over 2 – 3 minutes. Thereafter, at each cycle of therapy for a total of 4 days, the dose of 300 mg lidocaine was administered over 90 minutes. After the 2nd infusion, the patient was consistently pain-free. After 3 – 6 months the symptoms gradually reappeared but not in the severe intensity as prior to the start of lidocaine therapy. Save for a slight drop in blood pressure and a sensation of warmth in the whole body, there were no adverse reactions.
Discussion and review
 The diagnosis of Dercum´s disease is made clinically,
but histopathological examinations should be undertaken – especially to
rule out malignancy. Characteristically, painful subcutaneous adipose
tissue deposits occur in overweight females (Rubens figure) in middle age.
The patient treated by us was also overweight (80 kg, 158 cm). The
syndrome is 5 – 30 times more frequent in women than in men. The
majority of cases occur sporadically, as in the case presented. Only 3
publications describe a familial cluster based on autosomally dominant
inheritance with incomplete penetration [4, 5, 18]. There is no reliable
information on prevalence, since the pathogenesis is not completely
understood. Brorson and Fagher, however, estimate that in Sweden at least
10.000 persons have Dercum´s disease [3].
The diagnosis of Dercum´s disease is made clinically,
but histopathological examinations should be undertaken – especially to
rule out malignancy. Characteristically, painful subcutaneous adipose
tissue deposits occur in overweight females (Rubens figure) in middle age.
The patient treated by us was also overweight (80 kg, 158 cm). The
syndrome is 5 – 30 times more frequent in women than in men. The
majority of cases occur sporadically, as in the case presented. Only 3
publications describe a familial cluster based on autosomally dominant
inheritance with incomplete penetration [4, 5, 18]. There is no reliable
information on prevalence, since the pathogenesis is not completely
understood. Brorson and Fagher, however, estimate that in Sweden at least
10.000 persons have Dercum´s disease [3].
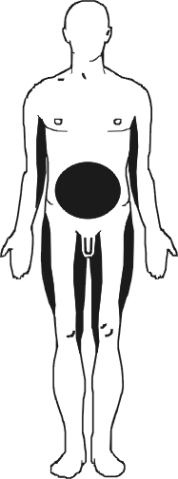
Tender and pressure-sensitive adipose tissue occurs particularly at various sites of predilection: the inner surface of the upper arm and elbow joint, abdomen, buttocks, the inner and outer surface of the thigh and knee joint (Fig. 2). As a rule, the face and the hands are spared. Usually the pain symptoms increase insidiously over months to years. Slight pressure, touch, closely fitting clothing or water when showering can be perceived as unpleasant. The pain is dependent on weather conditions and improves as a rule in dry heat. Our patient reported that the symptoms temporarily abated after a hot bath. According to the pain distribution pattern, the following sub-typing can be done [3]:
Type I (juxta-articular): painful fatty tumors at the inner surface of the knee, on the hips, less frequently at the elbow joint.
Type II (diffuse – generalized): diffuse tenderness on the dorsal surface of the upper arm, axillar, gluteal, on the abdomen, back and on the soles of the feet; abdominal pain can frequently predominate.
Type III (nodular): particularly lipomas painful on pressure; frequently also in the absence of overweight. Several of these lipomas were – as in the present case – histologically classified as angiolipomas, because they exhibited a pronounced density of blood vessels.
Dercum´s disease causes a number of psychosocial problems. The chronic hyperalgesia frequently impairs sexual relations or results in occupational invalidity. Emotional changes were repeatedly described in the literature such as, for example, irritability, depression, fatigue, lack of drive, sleep or memory disorders. These symptoms can also be interpreted as psychoreactive concomitant symptoms in the context of chronic pain symptoms (see the case reports of the American Self-help Group, Dercum's Support: http://www.dercum.org/). Many of our patient's symptoms described in the casuistics are typical accompanying symptoms of Dercum´s disease [3].
Swelling of the hands and fingers with accompanying paresthesias, numbness or carpal tunnel syndrome. Joint stiffness after rest, particularly in the morning. Dryness of the eyes and mouth. Telangiectasia with increased fragility of the dilated vessels; accordingly a tendency to bluish-red discoloration and ecchymoses; there is no disorder of coagulation. Headache (tension headache, migraine, cervicogenic headache).
Differential diagnoses
Other diseases coexist with Dercum´s disease, which result in a disorder of adipose tissue distribution and must therefore be considered in the differential diagnosis. For example, in benign symmetrical lipomatosis, adipose tissue pads occur, which in contrast with Dercum´s disease are not painful. The predominance is in middle-aged men. Fat accumulation in the area of the upper mediastinum can result in compression of the respiratory passages. According to Donhauser et al. [13] three types can be differentiated according to the localization of the fatty pads:
I. Madelung’s-disease [adenolipomatosis syndrome] or cervico-nuchal type;
II. Shoulder-girdle or pseudoathletic type:
III. Pelvic-girdle or gynecoid type.
An alcohol-abuse or hepatopathological etiology is assumed. Lipedema, in contrast, occurs in women with a genetically determined disorder of fat distribution. The underlying lipohypertrophy, wherein the extremities are thickened symmetrically with the trunk, predominantly affects the legs and is initially painless. Edema occurs in the course of lymphostasis due to the pressure of increased fat cells on small lymph vessels. Symptoms in the form of a feeling of heaviness, tension pain and pressure sensitivity or tenderness then occur. A collar-like termination of the edema over the ankle or wrist joint is typical [15].
Only in recent years has it become known that disorders of fat distribution can occur also in HIV patients in the context of retroviral therapy with protease inhibitors. This so-called HIV lipodistrophy is accompanied by atrophy of the adipose tissue in the face, extremities and buttocks. In contrast, there is an increase in adipose tissue on the trunk (truncal obesity) and neck (steer neck). A Cushing's habitus, which is frequently associated with metabolic disorders like hyperlipidemia, hypercholesterolemia and diabetes mellitus. As a rule, pain does not occur; however, cases with painful angiolipoma have been described [7].
Pathogenesis
The underlying pathogenetic mechanisms continue to be unknown. Furthermore, there are no systematic studies of larger numbers of cases and many Soviet and Swedish publications are difficult to access for western medicine.
Disorders of lipid metabolism or endocrine disorders are presumed to be the cause of the disease: Blomstrand et al. found a disorder of C18 fatty acids in two cases [2]. Fagher et al., however, arrived at a different conclusion in a study of 13 patients and found enlarged adipocytes at histological examination in comparison with controls of identical weights [14]. Histological studies have so far produced heterogeneous findings, wherein principally unremarkable adipose tissue was demonstrated. In 1888, Dercum found evidence of neuritis; this finding has, however, remained unconfirmed [9]. Until the mid 20th Century, the skin changes in lipomatosis were presumed to be in relation with myxedema. This was based on the frequently observed association of the pathologies with thyroid gland dysfunction, in particular hypothyroidism. When, however, in the 50s a female patient was treated with a thyroid hormone preparation and died, this hypothesis was abandoned [22]. Also in our patient, a subtotal resection of the thyroid was done in 1991 due to a nodular goiter and thereafter thyroxin was substituted. However, there was no hypothyrosis. It is theorized that subcutaneous adipose tissue deposits cause the pain symptoms by compression of the nerves. Skagen et al. demonstrated perturbations of subcutaneous blood flow in one female patient. Remission of pain and normalization of the subcutaneous blood flow for one month was achieved by systemic lidocaine administration. Based on this observation, the authors concluded an association of the sensitivity of the adipose tissue with a sympathetic dysregulation [21]. It is unclear, whether and if indeed, what role the increased blood vessel density of the angiolipoma plays in comparison with the lipomas. In a systematic study of 50 angiolipomas, Dixon et al. nevertheless found no connection between the extent of vascularization and pain intensity [12].
Therapy
Treatment of the Dercum´s disease has the primary goal of achieving relief from pain. Surgical excision or liposuction, for example of juxta-articular adipose tissue accumulations, can provide temporary pain relief. The effect diminishes over time and recurrences often occur [3]. We would therefore recommend surgical removal only of individual, particularly painful lipomas. Weight reduction appears not to have an affect on the pain symptomology and conventional analgesics have, as already mentioned, little effect.
Intravenous administration of lidocaine, on the other hand, provides in many patients weeks to months of complete freedom from pain. Accordingly, in the majority of publications, per treatment cycle single-dose approximately 5 mg / kg body weight of lidocaine is administered over 30 – 90 minutes [1, 11, 16, 20, 21]; in a case description, however, approximately 15 mg of lidocaine / kg body weight was administered i.v. on 4 –5 consecutive days, in order to achieve durable freedom from pain [17].
Systemic lidocaine administration has been used in pain therapy for some 40 years, especially in difficult to treat neuropathic pain. On the other hand, local administration of lidocaine is generally without effect. Alternatively or supplementally, the local anesthetic mexiletine, which can also be administered in tablet form. has been used successfully. Accordingly, dosages of 150 – 170 mg daily are administered [6, 8, 11, 20]. After consultation with the cardiology department of Medizinische Hochschule Hannover, no effort was made for intermediate treatment of our patient with mexiletine, since cardiovascular adverse effects must be taken into account. In view of the comparatively long persistence of the lidocaine effect in our patient, this risk was not justified. Prior to systemic lidocaine therapy, 2nd and 3rd degree AV block or sick sinus syndrome must be ruled out as contraindications. According to publications to date, tolerability of the treatment is generally good. During infusion, however, continuous ECG and RR monitoring should be done, because cardiovascular adverse effects must be taken into account. A fall in blood pressure, arrhythmias, bradycardia and, in very rare cases, asystole can occur. In addition, slight central nervous system side effects such as vertigo, dizziness or paresthesias can frequently occur. Many patients report slight headache or nausea after infusion. Furthermore, disorientation, vision and speech disturbances, tinnitus and tremor have been observed. Very rarely, reduction in vigilance, respiratory depression or epileptic events can occur.
The same contraindications apply to treatment with mexiletine and similar cardiovascular and central nervous system adverse events can occur. In addition, there are frequently gastrointestinal symptoms such as, for example, heartburn, abdominal pain, nausea and vomiting. Rare adverse effects are: skin reactions including Stevens-Johnson-syndrome, liver damage, pulmonary fibrosis, leukocytopenia or thrombocytopenia. In outpatient mexiletine treatment, therefore, weekly ECG and monthly prolonged ECG, as well and blood testing and liver values must be done.
Interestingly, the duration of the effect of lidocaine infusions (10 h to 12 months) is generally appreciably longer than one would have assumed based on the biological half-life (first 30 minutes after i.v. administration: half-life 7-10 min by tissue distribution; thereafter half-life 90 – 120 min by metabolism and excretion). The underlying mechanisms of activity are still incompletely understood. Animal experimental studies allow the hypothesis that an increased density of voltage-dependent sodium channels in damaged axons and in the associated spinal ganglia result in spontaneous ectopic discharges and thus to neuropathic pain. Electrophysiological studies show that by systemic lidocaine administration, these spontaneous discharges can be suppressed without blocking normal nerve conduction.
According to more recent studies, there are more than 10 different sodium channels that are responsible for the production and propagation of an action potential. Especially the sub-group of tetrodotoxin resistant sodium channels appears to play a special role in pain modulation. In peripheral nerve lesions down-regulation of the tetrodotoxin-resistant sodium flux and an up-regulation of the tetrodotoxin-sensitive sodium flux was demonstrated. Lidocaine acts approximately 4 times more potently on tetrodotoxin-sensitive sodium channels and was thus capable of correcting the imbalance of the sodium fluxes in neuropathic pain.
Summary
Pain in the area of the subcutaneous adipose tissue deposits is characteristic of the clinical picture of Dercum´s disease. Women in middle age are particularly affected by the disease. Unfortunately, the underlying pathogenetic mechanisms continue to be unknown. However, it is assumed that nociceptors and afferent axons are damaged by the local compression by the adipose tissue deposits. In addition, a sympathetic dysregulation and perturbation of the subcutaneous blood flow appears to be of significance. Chronic pain (persisting longer than 3 months) occurs and appears spontaneously or can be provoked by light contact stimuli (allodynia). Cold external temperatures also trigger the occurrence of the pain described as burning, cutting or stabbing. The symptoms are hardly affected by conventional analgesics or classical pain modulating therapy. Based on the stated features, we consider the assignment of the disease to the neuropathic pain syndromes as justified. Dercum´s disease appears to respond satisfactorily to systemic lidocaine or mexiletine administration. This therapy has been shown in the past to be effective in numerous other neuropathic pain syndromes.
The original articel can be downloaded: NERVENARZT
Literature:
1. Atkinson RL (1982) Intravenous lidocaine for the treatment of intractable pain of Adiposis dolorosa. Int J Obes 6:351–357
2. Blomstrand R, Juhlin L, Nordenstam H, Ohlsson R, Werner B, Engstrom J (1971) Adiposis dolorosa associated with defects of lipid metabolism. Acta Derm Venereol 51:243–250
3. Brorson H, Fagher B (1996) Dercum's disease. Fatty tissue rheumatism caused by immune defense reaction? Lakartidningen 93:1430–1436
4. Campen R, Mankin H, Louis DN, Hirano M, Maccollin M (2001) Familial occurrence of adiposis dolorosa. J Am Acad Dermatol 44:132–136
5. Cantu JM, Ruiz-Barquin E, Jimenez M, Castillo L, Macotela-Ruiz E (1973) Autosomal dominant inheritance in adiposis dolorosa (Dercum's disease). Humangenetik 18:89–91
6. Chabal C, Jacobson L, Mariano A, Chaney E, Britell CW (1992) The use of oral mexiletine for the treatment of pain after peripheral nerve injury. Anesthesiology 76:513–517
7. Dank JP, Colven R (2000) Protease inhibitorassociated angiolipomatosis. J Am Acad Dermatol 42:129–131
8. Dejgaard A, Petersen P, Kastrup J (1988) Mexiletine for treatment of chronic painful diabetic neuropathy. Lancet 29:9–11
9. Dercum FX (1888) A subcutaneous connective tissue dystrophy of the arms and back, associated with symptoms resembling Myxoedema. Univ Med Mag Philadelphia 1:140–150
10. Dercum FX (1892) Three cases of a hitherto unclassified affection resembling in its grosser aspects obesity, but associated with special nervous symptoms – adiposis dolorosa. Am J Med Sci 104:521–535
11. Devillers AC, Oranje AP (1999) Treatment of pain in adiposis dolorosa (Dercum's disease) with intravenous lidocaine: a case report with a 10-year follow-up. Clin Exp Dermatol 24:240–241
12. Dixon AY, McGregor DH, Lee SH (1981) Angiolipomas: an ultrastructural and clinicopathological study. Hum Pathol 12:739–747
13. Donhauser G, Vieluf D, Ruzicka T, Braun-Falco O (1991) Benigne symmetrische Lipomatose Launois-Bensaude Typ III und Bureau-Barrière- Syndrom. Hautarzt 42:311–314
14. Fagher B, Monti M, Nilsson-Ehle P, Akesson B (1991) Fat-cell heat production, adipose tissue fatty acids, lipoprotein lipase activity and plasma lipoproteins in adiposis dolorosa. Clin Sci 81:793–798
15. Herpertz U (1995) Das Lipödem. Z Lymphologie 19:1–7
16. Iwane T, Maruyama M, Matsuki M, Ito Y, Shimoji K (1976) Management of intractable pain in adiposis dolorosa with intravenous administration of lidocaine. Anesth Analg 55:257–259
17. Juhlin L (1986) Long-standing pain relief of adiposis dolorosa (Dercum's disease) after intravenous infusion of lidocaine. J Am Acad Dermatol 15:383–385
18. Lynch HT, Harlan WL (1963) Hereditary factors in adiposis dolorosa (Dercum's disease). Am J Hum Genet 15:184–190
19. Mao J, Chen LL (2000) Systemic lidocaine for neuropathic pain relief.Pain 87:7–17
20. Petersen P, Kastrup J (1987) Dercum's disease (adiposis dolorosa).Treatment of the severe pain with intravenous lidocaine. Pain 28:77–80
21. Skagen K, Petersen P, Kastrup J, Norgaard T (1986) The regulation of subcutaneous blood flow in patient with Dercum's disease. Acta Derm Venereol 66:337–339
22. Steiger WA, Litvin H, Lasche EM, Durant TM (1952) Adiposis dolorosa (Dercum's Disease). NEJM 247:393–396
Thanks to Dr. Steiner for permission to display this article.
Return to the List of Articles
Return to the
Dercum's Disease Home Page
Last updated 29 July 2009
Comments about the web page format
should be sent to the Don
Please don't forget that the information provided on this site is designed to support, not replace, the relationship that exists between a patient and his or her physician.